CD-19 CAR-T Cell Therapy in Indolent Lymphoma: Five-Year Follow-Up Results from ZUMA-5
Long-term data from the ZUMA-5 trial demonstrate durable responses with axicabtagene ciloleucel, offering new hope for patients with relapsed or refractory follicular lymphoma.
The Challenge of Relapsed Indolent Lymphoma
For patients with follicular lymphoma who relapse after multiple lines of therapy, treatment options have historically offered diminishing returns. While indolent non-Hodgkin lymphomas often respond initially to chemoimmunotherapy, each subsequent relapse typically brings shorter remissions and accumulating toxicities. Patients who progress within 24 months of their first anti-CD20 therapy—a group known as POD24—face particularly poor outcomes with conventional approaches.
This clinical reality has driven interest in whether CAR T-cell therapy, which has transformed outcomes in aggressive lymphomas, might offer similar benefits in indolent disease. The ZUMA-5 trial set out to answer that question, and after more than five years of follow-up, the results provide meaningful clarity.
Understanding the Therapeutic Approach
Axicabtagene ciloleucel (axi-cel) is an autologous anti-CD19 chimeric antigen receptor T-cell therapy. The treatment involves collecting a patient's own T cells, genetically engineering them to recognize and attack CD19-expressing cancer cells, expanding them in a manufacturing facility, and infusing them back into the patient following lymphodepleting chemotherapy.
What distinguishes CAR T-cell therapy from conventional treatments is its potential for durable disease control through a single infusion. Unlike chemotherapy or antibody therapies that require repeated dosing, CAR T cells can persist in the body and provide ongoing immune surveillance against residual disease.
Study Design and Patient Population
ZUMA-5 enrolled 159 patients with relapsed or refractory indolent non-Hodgkin lymphoma, including 127 with follicular lymphoma and 31 with marginal zone lymphoma. This was a heavily pretreated population: patients had received a median of three prior therapies, 64% had received three or more prior lines, and 71% were refractory to their most recent treatment.
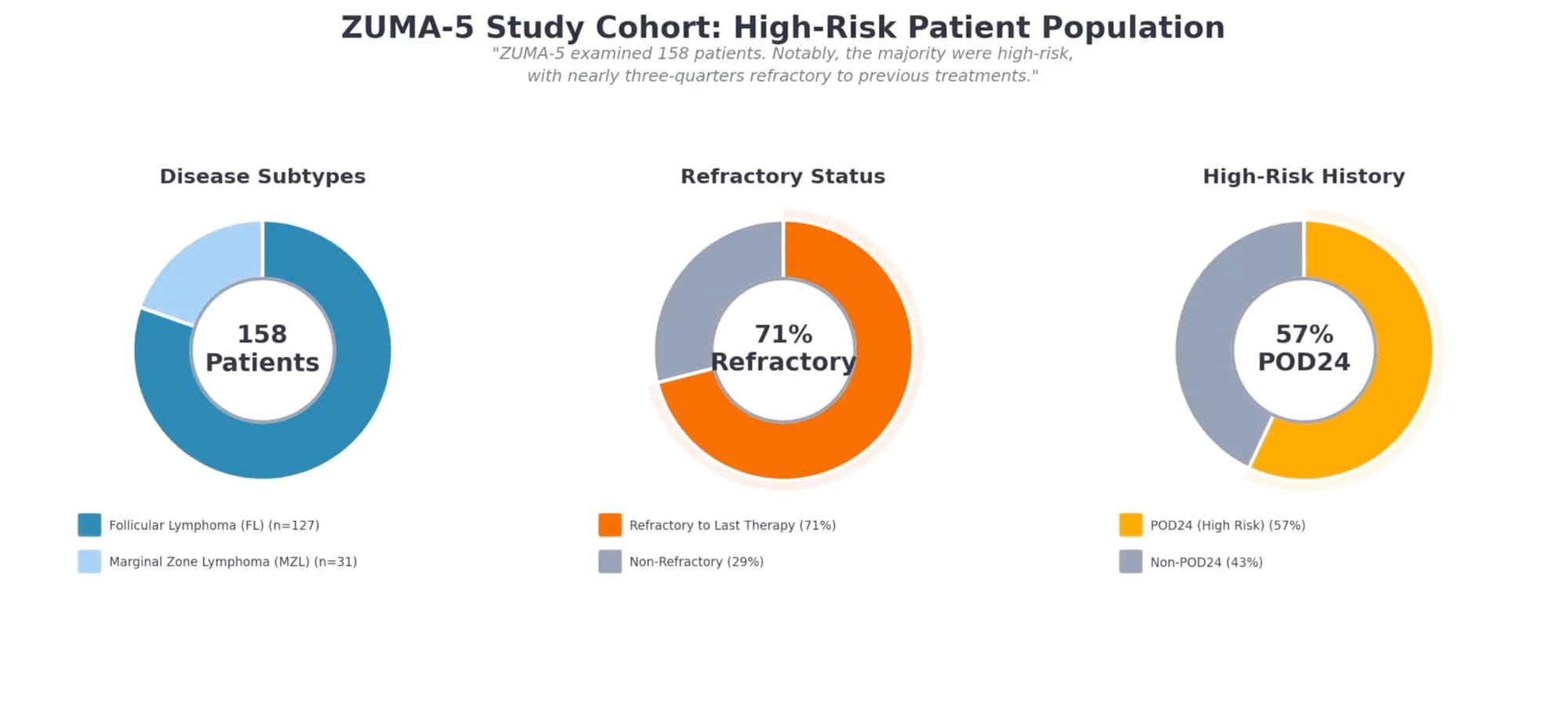
Notably, 57% of patients had experienced POD24—early progression after initial anti-CD20 therapy—a characteristic associated with inferior outcomes. More than half had high tumor bulk by GELF criteria, and 87% had advanced-stage disease. This patient population represents those with the greatest unmet need: individuals for whom standard options had already failed.
Of the 159 enrolled patients, 152 received axi-cel infusion at a target dose of 2 × 10⁶ CAR T cells/kg following fludarabine and cyclophosphamide conditioning.
Efficacy: Durable Responses Over Five Years
After a median follow-up of 64.6 months—more than five years—the efficacy data demonstrate sustained benefit that extends well beyond what is typically seen with conventional therapies.
Response rates remained consistent with earlier analyses:
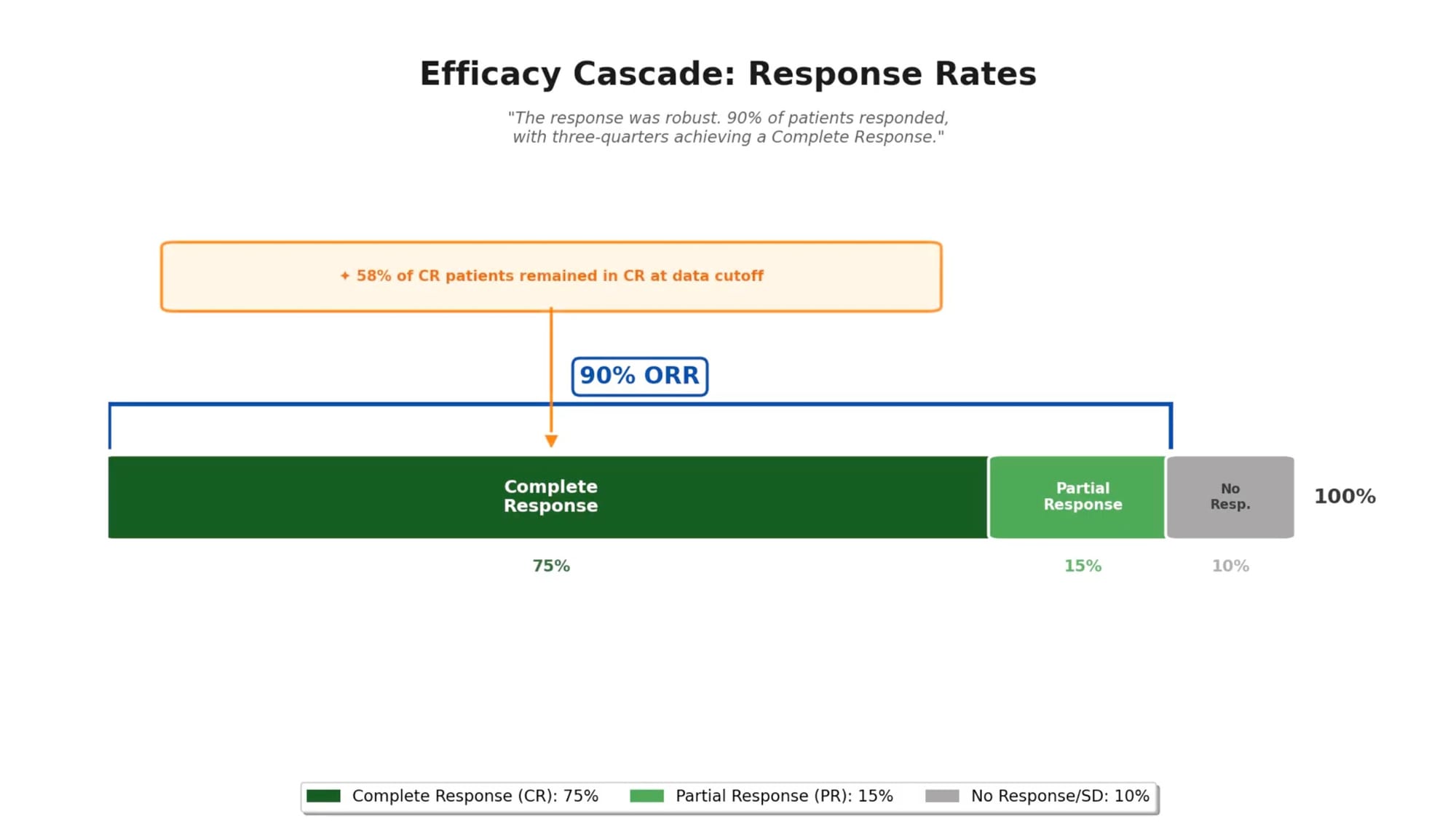
- Overall response rate: 90%
- Complete response rate: 75%
- Median time to best response: 1.0 month in follicular lymphoma
Durability proved to be the defining feature of these results:
- Median duration of response: 60.4 months
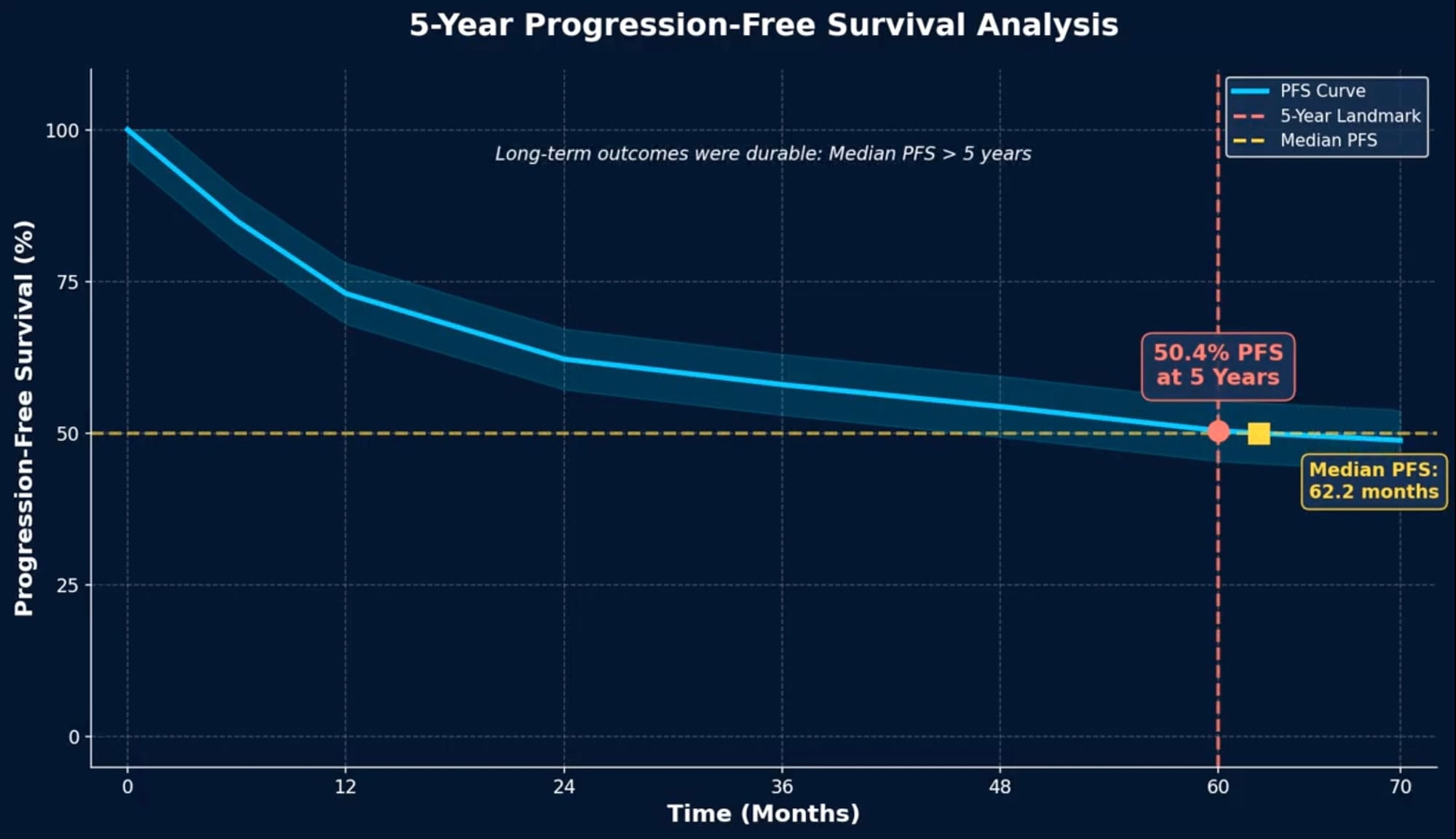
- Median progression-free survival: 62.2 months
- 50.4% of patients reached the 60-month progression-free survival landmark
- 58% of patients who achieved complete response remained in complete response at data cutoff
Perhaps most striking: at data cutoff, 55% of patients were alive without having required any subsequent anticancer therapy. For a population where the median prior therapies numbered three and the majority had refractory disease, this represents a meaningful departure from the expected trajectory.
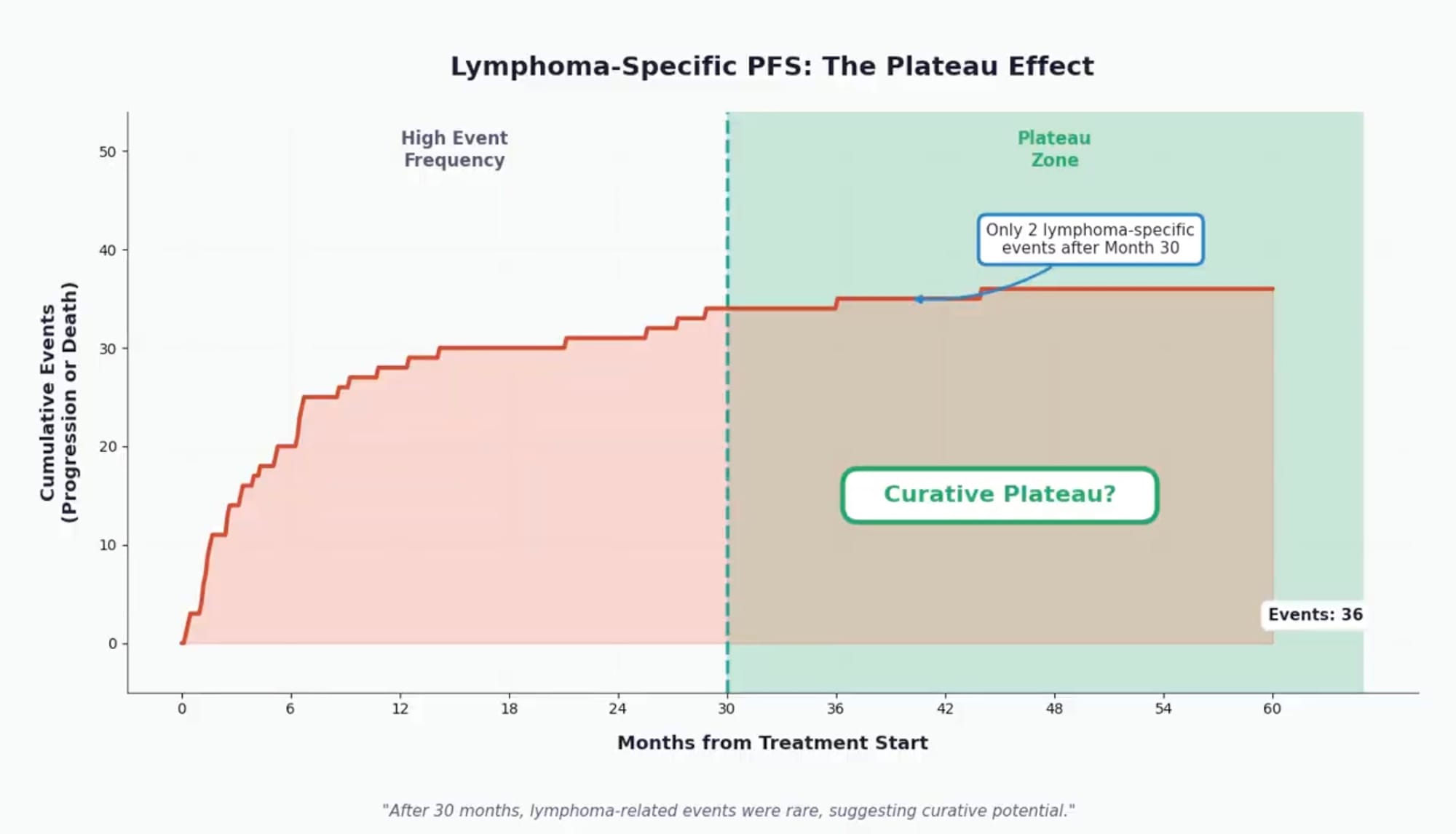
A plateau in lymphoma-specific outcomes: The investigators observed that after 30 months post-infusion, progression or lymphoma-related deaths became rare—only two such events occurred beyond this timepoint. This plateau suggests that patients who remain in remission at 2.5 years have a high likelihood of sustained disease control, a pattern consistent with potential curative benefit for a subset of patients.
For patients who relapsed and were retreated with axi-cel (median follow-up of 45.5 months post-retreatment), the median duration of response was 11.6 months, with 38% maintaining ongoing response—demonstrating that retreatment remains an option for some.
Safety: A Manageable Long-Term Profile
Long-term safety data are essential for any therapy intended to provide lasting benefit. In ZUMA-5, the five-year follow-up revealed no new safety signals beyond what was observed in earlier analyses.
Mortality outcomes over time: Over the follow-up period, 46 patients (30%) died. The causes of death illuminate the long-term risk profile:
- Progressive disease accounted for 14 deaths (9%)
- Nine additional deaths (6%) occurred after disease progression but from other causes
- Second primary malignancies: 6 deaths (4%)
- Infection-related deaths: 11 (7%)
- Cardiac-related deaths: 3 (2%)
The temporal pattern of deaths is informative. Most occurred within the first three years, with only four deaths in years four and five combined—and only one death after year five. Late-onset toxicities were infrequent and largely unrelated to axi-cel treatment.
Second primary malignancies were observed, some considered potentially related to lymphodepleting chemotherapy and/or axi-cel, though these occurred rarely. This finding aligns with observations across CAR T-cell programs and warrants ongoing surveillance.
Factors Associated with Durable Benefit
Correlative analyses identified biological features associated with prolonged response and survival:
- Robust early CAR T-cell expansion: Patients whose CAR T cells proliferated vigorously in the early post-infusion period experienced better outcomes
- Naïve T-cell phenotype in the product: Products enriched for less-differentiated T cells were associated with more durable responses
These findings provide mechanistic insight into why some patients experience exceptional durability and may inform future efforts to optimize manufacturing or patient selection.
Interpreting the Results in Context
How should clinicians and patients interpret these five-year data? Several considerations are important.
Comparison to historical benchmarks: While cross-trial comparisons require caution, the median progression-free survival of 62.2 months in ZUMA-5 compares favorably to outcomes typically seen with salvage chemoimmunotherapy in relapsed follicular lymphoma, where median PFS generally ranges from 12 to 24 months depending on the regimen and patient selection.
The plateau phenomenon: Unlike in diffuse large B-cell lymphoma where a clear survival plateau emerges around two years post-CAR T, the pattern in follicular lymphoma is more gradual—partly because of the longer latency before progression events manifest clinically and competing risks of death from other causes. Lymphoma-specific progression-free survival may more accurately capture the curative potential in this disease.
Acknowledging limitations: ZUMA-5 was a single-arm study, and outcomes should be interpreted in that context. Patient selection, supportive care at specialized centers, and the specific characteristics of enrolled patients all influence results. Not all patients in routine practice will match the trial population.
What This Means for Patients and Clinicians
For patients with relapsed or refractory follicular lymphoma who have exhausted multiple prior therapies, these data offer a realistic basis for hope. A therapy that can produce durable remissions lasting five years or longer—with more than half of patients alive and treatment-free at data cutoff—represents a meaningful advance.
For treating physicians, the findings support consideration of axi-cel for appropriate patients, particularly those with POD24 or multiply relapsed disease where conventional options offer limited durability. The manageable long-term safety profile, absence of new late toxicity signals, and the correlation between early CAR T-cell expansion and durable benefit all inform clinical decision-making.
Looking Ahead
The ZUMA-5 five-year follow-up adds to a growing body of evidence that CAR T-cell therapy can fundamentally alter the disease course for patients with indolent lymphoma. Ongoing research continues to explore optimization strategies, earlier use in the treatment sequence, and approaches to enhance the durability of responses for patients who relapse.
For a disease historically characterized by repeated remissions and relapses, the emergence of a plateau in lymphoma-specific outcomes—with rare events beyond 30 months—suggests that for a substantial proportion of patients, a single CAR T-cell infusion may provide lasting disease control.
This case study summarizes findings from: Neelapu SS, et al. "Five-Year Follow-Up Analysis of ZUMA-5: Axicabtagene Ciloleucel in Relapsed/Refractory Indolent Non-Hodgkin Lymphoma." Journal of Clinical Oncology. 2025;43(33). Clinical trial: NCT03105336. Study supported by Kite, a Gilead Company.